On January 2, 2026, the Feng Yue/Zhang Yi team at Beijing University of Chemical Technology, in collaboration with the Zhang Kaiming team from the School of Biomedical Sciences at the University of Science and Technology of China, published a research paper titled “Structural Insights into Type-I and Type-II Lamassu Antiphage Systems” in Nature Chemical Biology. The study reports high-resolution cryo-EM structures of multiple states within Type-I and Type-II Lamassu systems. Combined with in vitro biochemical and phage defense experiments, it reveals their unique assembly mechanisms and elucidates how this “cellular autophagy” switch is precisely regulated. This marks Professor Feng Yue's fourth paper published in Nature Chemical Biology as corresponding author.
On the microscopic battlefield, bacteria and invading viruses (bacteriophages) engage in an endless “arms race.” To resist phage infection, bacteria have evolved highly complex defense networks, including an extreme strategy known as abortive infection (Abi). This mechanism sacrifices individual cells for the collective good: upon phage infection, bacteria initiate a program leading to their own death or cessation of growth, effectively destroying themselves along with the virus to prevent replication and spread to neighboring cells. Statistics indicate that over 70% of prokaryotes possess this defense strategy. Among the myriad Abi mechanisms, an anti-phage system named Lamassu was first identified by scientists in 2018. This system exists in two variants (Type I and Type II), exhibiting remarkable diversity by activating defenses through recognition of specific DNA sequences (such as palindromic sequences) in invaders. However, despite scientists having predicted the functions of individual Lamassu components, the precise assembly and operation of this intricate microscopic machinery remains a significant enigma.
In this study, the collaborative team first discovered that the core of the Lamassu system is a dimeric scaffold composed of the SMC (Structural Maintenance of Chromosomes) family protein LmuB, forming a giant pair of “clamps.” In the Type II system, this pair also incorporates a unique LmuC component positioned at the complex's center. During “peacetime”—when no viral invasion occurs (the quiescent state)—the nuclease LmuA, responsible for the killing function, is tightly “locked” as a monomer onto the helical domain of LmuB. This conformation isolates LmuA and maintains its inactive state, akin to engaging a weapon's safety lock to prevent accidental damage to the host bacterium's own DNA. The collaborative team subsequently resolved the structures of both Lamassu systems bound to DNA substrates. Combined with in vitro DNA cleavage experiments, they discovered that unlike traditional SMC complexes that wrap around DNA, the Lamassu system detects danger by directly binding to the “ends” of double-stranded DNA. These DNA ends typically appear in large quantities only during massive phage replication or when bacterial genomes are damaged. The team also discovered an intriguing phenomenon: high ATP concentrations (indicating cellular health) suppress the system's nuclease activity, while low ATP levels (representing energy depletion due to phage attack) promote DNA binding and cleavage. This reveals a dual-check mechanism for sensing DNA ends and monitoring energy levels. The collaborative team also captured the release and polymerization of effector proteins: phage DNA binding triggers the release of previously “imprisoned” LmuA monomers. These liberated monomers rapidly deform within the cytoplasm and assemble into active tetrameric structures. This tetramer functions like a high-speed shredder, indiscriminately cleaving all cellular DNA, ultimately causing the infected bacterium's death (aborted infection). This self-sacrifice blocks further phage spread within the bacterial population.
In summary, this study not only presents a comprehensive molecular portrait of the Lamassu system from its quiescent state to activation but also uncovers a novel function of SMC family proteins in prokaryotic immunity—serving not merely as chromosome maintainers but as the core scaffold of the immune system. By deciphering the activation pathway of LmuA from monomer to tetramer, it provides a new theoretical framework for understanding how bacteria utilize polymerization mechanisms to regulate immune responses.
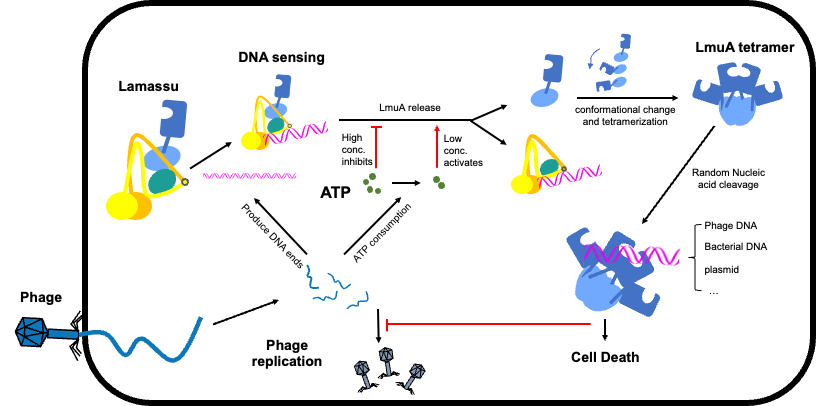
Figure 1 Schematic of the bacterial Lamassu defense system
Li Ming, PhD candidate at the University of Science and Technology of China (USTC), and postdoctoral researcher Zhao Xiaolong, along with PhD candidates Zhao Xingyu, Li Dong, and Xiong Weijia from Beijing University of Chemical Technology (BUCT), are the co-first authors of this paper. Professor Feng Yue and Associate Professor Zhang Yi from the School of Life Sciences and Technology at BUCT, and Professor Zhang Kaiming from the School of Life Sciences and Medicine at USTC, are the co-corresponding authors.
Original article link:
https://www.nature.com/articles/s41589-025-02102-z
About Professor Feng Yue:
Feng Yue is a professor and doctoral advisor at Beijing University of Chemical Technology. He received his Ph.D. from Tsinghua University in 2013 and joined Beijing University of Chemical Technology the same year. For years, Feng Yue's research group has employed biochemical and molecular biological, structural biological, and cellular biological approaches to investigate the structure and function of proteins involved in microbial-host immune interactions. The group has published 36 SCI papers as corresponding authors (including co-corresponding authors), appearing in Nature (2018, 2024), Cell (2023, 2025), Mol Cell (2020, 2022, 2024), Nat Chem Biol (2022, 2024, 2025a, 2025b), Nat Commun (2020, 2022), and PNAS (2024). Awards include: National Outstanding Young Professional (2020), Beijing Young Scholar (2024), 19th Fok Ying Tung Foundation Young Scientist Award (2024), Beijing Outstanding Young Talent (2020), Second Prize of the Chinese Medical Science and Technology Award (2024), China's Top Ten Emerging Science and Technology Figures (2018), Beijing Science and Technology Rising Star (2019), and Outstanding Instructor Award in the Beijing Higher Education Institutions Young Teachers Teaching Fundamentals Competition (2023).


