On March 18, 2022, Professor Feng Yue's research group at our School of Life Sciences published a research paper titled "AcrIF5 specifically targets DNA-bound CRISPR-Cas Surveillance Complex for inhibition" online in the journal "Nature Chemical Biology." The paper reports the cryo-EM structure of Csy-dsDNA-AcrIF5 and the crystal structure of the AcrIF5 protein, revealing the molecular mechanism by which AcrIF5 inhibits the type I-F CRISPR-Cas system.

In 2020, the Feng Yue research group discovered that AcrIF5 could not stably bind to Csy or Cas2/3, suggesting that it likely inhibits the I-F type CRISPR-Cas system in a unique way. In this study, the Feng Yue research group first determined the binding of AcrIF5 to Csy-dsDNA using multiple methods, prepared the complex sample of Csy-dsDNA-AcrIF5, and in collaboration with Tsinghua University's Yang Maojun research group, elucidated the structure of this complex using cryo-electron microscopy, and also resolved the crystal structure of AcrIF5. Interestingly, in the complex structure, AcrIF5 binds to the "abdomen" region of the Csy complex, composed of the Cas7f subunit, in the form of a pentamer, while the electron density of the portion of Cas8f subunit responsible for recruiting Cas2/3 nuclease (Cas8f-HB) disappears. Upon further structural analysis, they found that AcrIF5.3/5.4 could cause severe steric hindrance with the Cas8f-HB of Csy-dsDNA, leading them to speculate that AcrIF5 might exert its inhibitory effect by converting the “locked state” of Cas8f-HB responsible for recruiting Cas2/3 nuclease to a "freely swinging state". Subsequent Pull-down and Cas2/3 recruitment experiments confirmed that AcrIF5 could indeed compete with Cas8f-HB to bind the main body of Csy-dsDNA, thereby inhibiting the recruitment of Cas2/3, thus proposing the working mechanism of AcrIF5 (Figure 2a). In addition, the research group's bioinformatics analysis of 32 known organisms containing the AcrIF5 gene revealed that the AcrIF5 gene does not exist alone but coexists with genes such as AcrIF3. Combined with the Cas2/3 recruitment experiments, they proposed a collaborative model where in the process of inhibiting Cas2/3 recruitment, AcrIF3 takes the lead, and AcrIF5 supplements the functional role of AcrIF3 (Figure 2b).
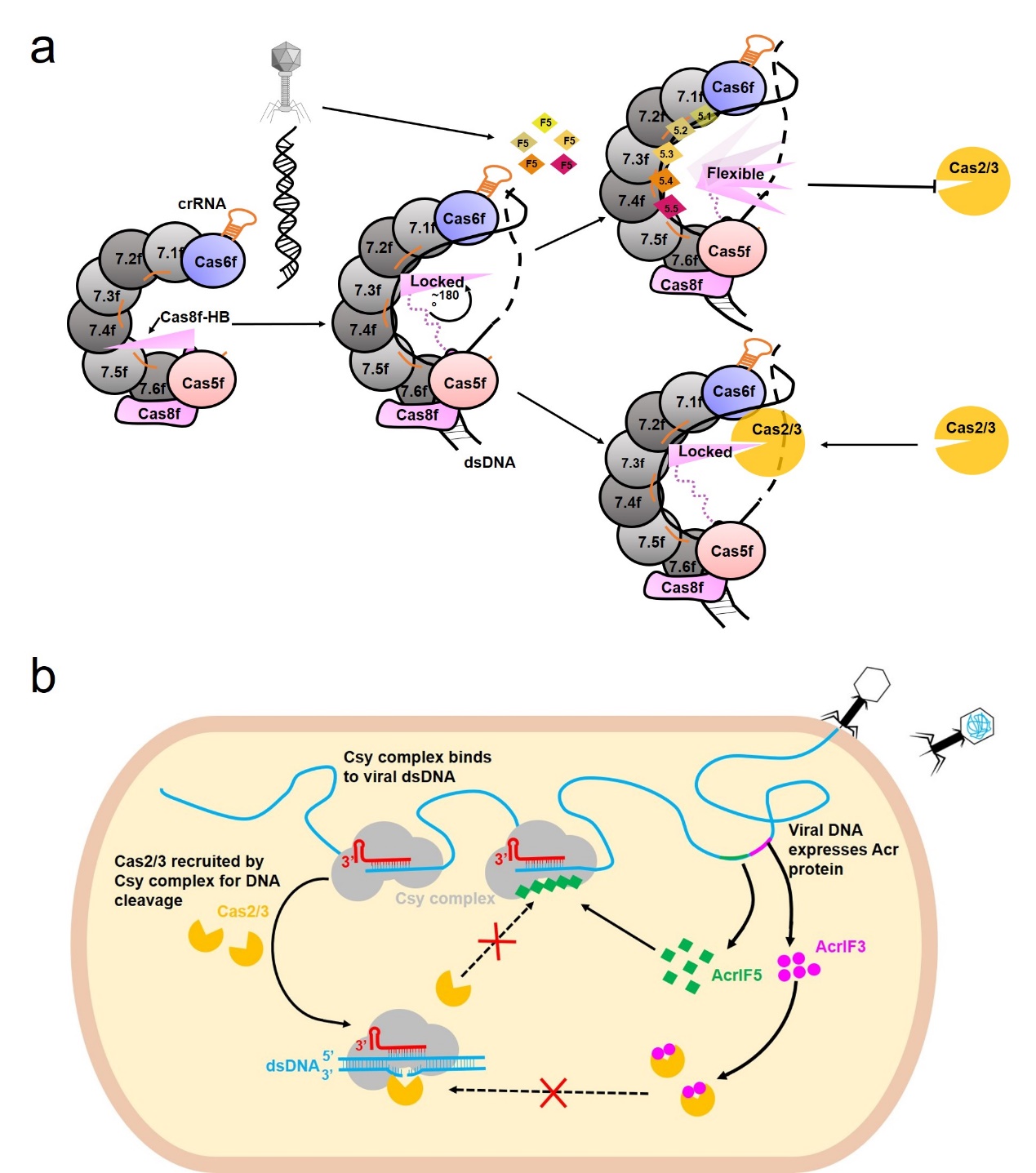
Figure 1: Mechanism of action of AcrIF5
Beijing University of Chemical Technology Ph.D. student Jie Yongchao, Tsinghua University postdoctoral fellow Zhang Laixing, Beijing University of Chemical Technology master student Gao Zhengyu are the co-first authors of this paper. Professor Feng Yue is the corresponding author of this paper, and Beijing University of Chemical Technology is the first completion unit. Professor Yang Maojun from Tsinghua University provided a lot of guidance in electron microscope structure analysis, and the collection of electron microscope data and crystal diffraction data received strong support from the Southern University of Science and Technology Cryo-EM Center and the Shanghai Synchrotron Radiation Facility.
Original Link: https://www.nature.com/articles/s41589-022-00995-8
Corresponding Author's Bio:
Feng Yue, professor at the School of Life Sciences, Beijing University of Chemical Technology, doctoral supervisor, recipient of the National Excellent Youth Fund (2018). Mainly engaged in the study of molecular mechanisms of pathogen-host interactions using disciplines such as structural biology, biochemistry, and cell biology. Have published 38 SCI papers, of which 16 were published as corresponding author in journals such as "Nature," "Nat. Chem. Biol.," "Mol. Cell," "Nat. Plants," and "Nat. Commun." Led multiple national and provincial projects as the principal investigator. Honors include National Young Skilled Talent (2020), Beijing Excellent Young Talent (2020), Beijing Science and Technology Progress Award Second Prize (2018), China's Top Ten Emerging Science and Technology Figures (2018), and Beijing Science and Technology Rising Star (2019).
