
First Author: Fei An
Corresponding Authors: Liang Hao, Liu Jianzhang
Affiliated Institutions: Peking University School of Stomatology; Beijing University of Chemical Technology
Research Highlights
Ensuring a stable soft tissue seal in the transmucosal region of titanium abutments is critical for the long term success of dental implants; however, conventional titanium abutment surfaces lack inherent antibacterial functions. Herein, a multifunctional antibacterial coating was developed by immobilizing pH responsive nanorods composed of tanshinone IIA, lactoferrin, and sodium alginate onto a polydopamine modified titanium surface. The resulting Ti&PDA@NRs coating achieved high tanshinone IIA loading through synergistic electrostatic adsorption and covalent crosslinking. The coating exhibited pH modulated release behavior, characterized by accelerated tanshinone IIA release under acidic conditions and sustained release at physiological pH. Antibacterial evaluations against representative oral bacterial strains demonstrated that the coating effectively inhibited bacterial adhesion and biofilm formation via surface contact killing, while the released tanshinone IIA further suppressed planktonic bacteria, confirming its dual mode antibacterial mechanism. Mechanistic studies revealed that the antibacterial activity was associated with disruption of bacterial membrane structures and elevation of intracellular reactive oxygen species levels. Cytocompatibility assessments using human gingival fibroblasts (HGFs) and mouse NIH/3T3 cells under both normal and inflammatory conditions confirmed low cytotoxicity. Moreover, the coating maintained good stability and sustained tanshinone IIA release performance under simulated oral environments. Collectively, this study presents a clinically relevant antibacterial surface strategy to address bacteria related challenges in the transmucosal region of implants.
Key Points Analysis
Key Points Overview: This study aims to address the critical clinical challenge of the lack of inherent antibacterial functionality on traditional titanium abutment surfaces. The core strategy involves constructing an intelligent antibacterial coating capable of “on demand release” in response to changes in the oral microenvironment. The coating system ingeniously combines the natural antibacterial component tanshinone IIA with the biocompatible carriers lactoferrin and sodium alginate to form pH responsive nanorods via self assembly. Utilizing the strong adhesion and chemical reactivity of polydopamine, these nanorods are stably integrated onto the titanium abutment surface to form a Ti&PDA@NRs composite coating.
A key physicochemical characteristic of the coating is its intelligent release behavior. Under pH 5.0 conditions, which simulate the acidic environment caused by bacterial biofilm metabolism, the nanorod structures within the coating respond, significantly accelerating the release rate of tanshinone IIA to achieve differential antibacterial action at infection sites. Under pH 7.4 conditions, which simulate the physiological environment of healthy tissues, the drug maintains a slow, sustained release, providing long term preventive protection. This differentiated release pattern effectively balances immediate antibacterial needs with long term maintenance requirements. Furthermore, stability tests indicated that the coating maintains good structural integrity and sustained drug release performance under simulated oral conditions involving moisture, micromotion, and pH fluctuations, meeting the basic durability requirements for clinical application.
Antibacterial performance evaluation constitutes a major part of this study. The effectiveness of the coating was comprehensively validated through in vitro experiments against representative oral bacteria, namely Gram positive Streptococcus mutans and Gram negative Porphyromonas gingivalis. Results showed that the coating strongly inhibited initial bacterial adhesion on the material surface, significantly killed already attached bacteria, and fundamentally suppressed the formation and maturation of complex biofilms. Its antibacterial mechanism was confirmed as an efficient “dual mode” synergy: first, the nanostructure of the coating surface and its loaded active components directly damage bacterial cells through contact; second, tanshinone IIA molecules continuously released from the coating diffuse into the surrounding environment to eliminate planktonic bacteria. This combination of “surface killing” and “release killing” establishes a three dimensional, multi level antibacterial defense.
To deeply understand the antibacterial principle, the research team conducted systematic mechanistic investigations. Scanning electron microscopy images clearly showed that bacterial cells in contact with the coating underwent severe morphological distortion, with cell membrane rupture and pore formation. Corresponding biochemical assays further confirmed the leakage of intracellular macromolecules such as proteins and nucleic acids, indicating disruption of cell membrane permeability and loss of integrity. Concurrently, coating treatment induced a sharp rise in intracellular reactive oxygen species levels in bacteria, accompanied by a rapid decline in ATP content, suggesting that the coating induces strong oxidative stress and interferes with normal bacterial energy metabolism. The combination of physical damage to the cell membrane and internal chemical stress ultimately leads to bacterial death.
For any biomaterial intended for human use, safety is a primary consideration. This study rigorously evaluated the biocompatibility of the coating at the in vitro cellular level. Human gingival fibroblasts, relevant to oral soft tissue healing, and a standard mouse embryonic fibroblast cell line were selected for testing under both normal culture conditions and simulated inflammatory conditions induced by lipopolysaccharide. A series of experimental results, including cell viability assays, cell morphology observation, live/dead cell staining, and measurement of lactate dehydrogenase release as a marker of cell damage, consistently demonstrated that compared with bare titanium surfaces, the Ti&PDA@NRs coating exhibited no significant cytotoxicity. Cells were able to adhere normally and maintain proliferative activity on the coating surface. Even under inflammatory conditions, the coating did not exacerbate cell damage, fully confirming its good biological safety.
In summary, this study successfully developed and systematically validated an intelligent, responsive antibacterial coating for titanium abutments based on natural active components. Through sophisticated design, this coating not only achieves pH regulated drug release and dual mode antibacterial action with excellent and durable anti biofilm properties but also demonstrates reliable safety at the cellular level. This achievement provides an innovative solution with a clear mechanism, comprehensive performance, and great potential for clinical translation for proactively preventing soft tissue seal failure and peri implantitis in the transmucosal region of implants caused by bacterial infection, from the perspective of material surface functionalization.
Graphical Abstract
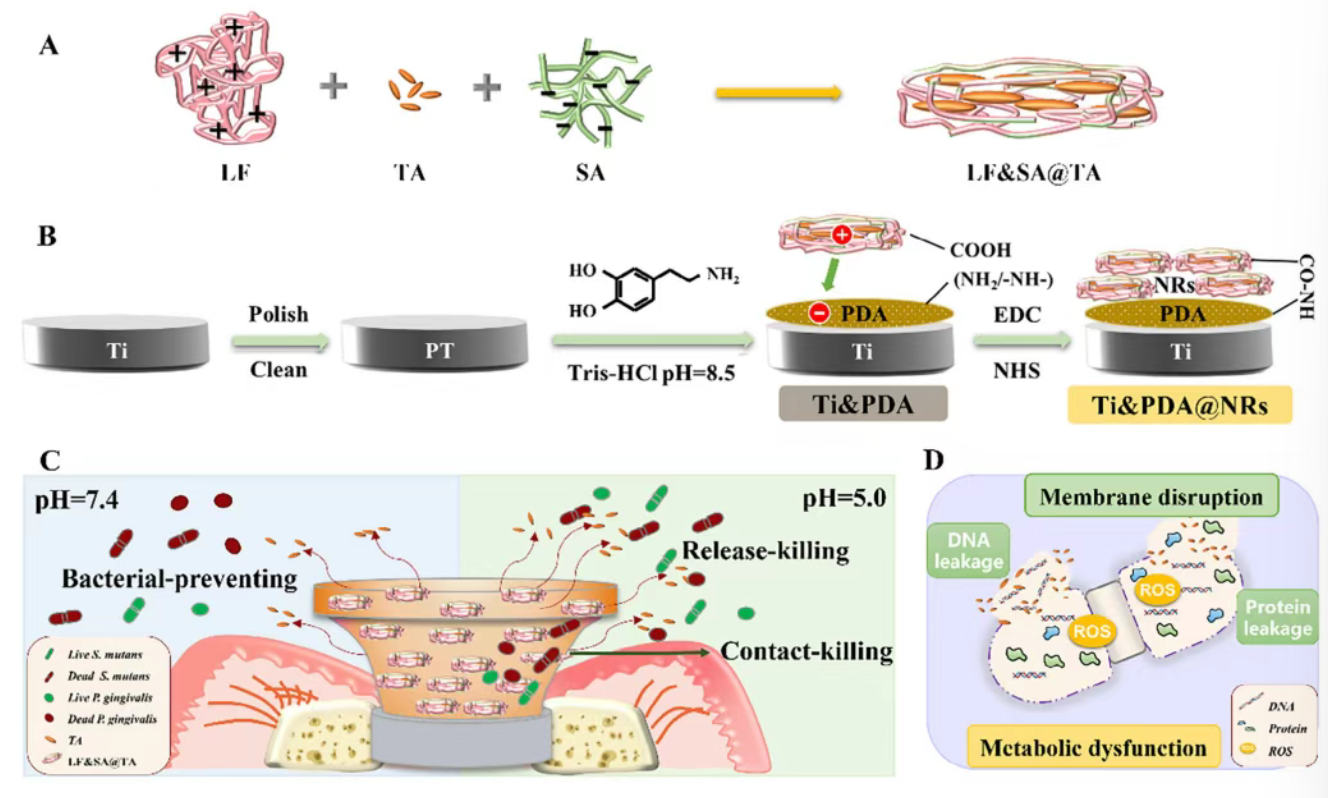
Figure 1. (A) Construction process of LF&SA@TA nanorods. (B) Fabrication procedure of Ti&PDA@NRs coating through electrostatic adsorption and covalent coupling. (C) Antibacterial properties of LF&SA@TA nanorods and Ti&PDA@NRs coating. (D) Schematic illustration of the antibacterial mechanism of Ti&PDA@NRs coating against Streptococcus mutans.
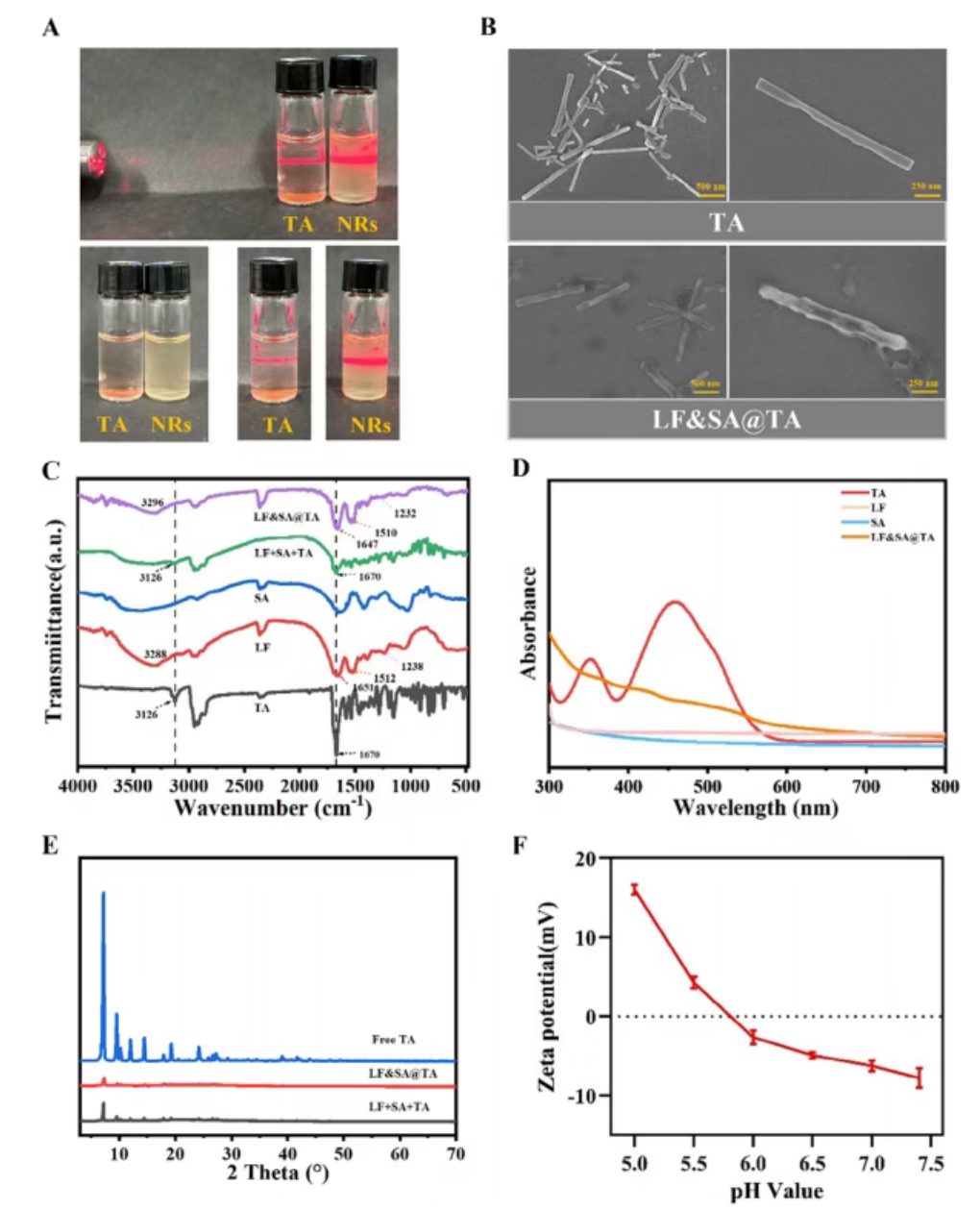
Figure 2. Characterization of LF&SA@TA nanorods. (A) Representative digital photographs of TA and LF&SA@TA nanorods. (B) Scanning electron microscopy images of TA and LF&SA@TA nanorods. (C) FTIR spectra of free TA, LF, SA, physical mixture of LF+SA+TA, and LF&SA@TA nanorods. (D) UV-Vis absorption spectra of free TA, LF, SA, and LF&SA@TA nanorods. (E) XRD patterns of free TA, physical mixture of LF+SA+TA, and LF&SA@TA nanorods. (F) Zeta potential values of LF&SA@TA nanorods at different pH values.
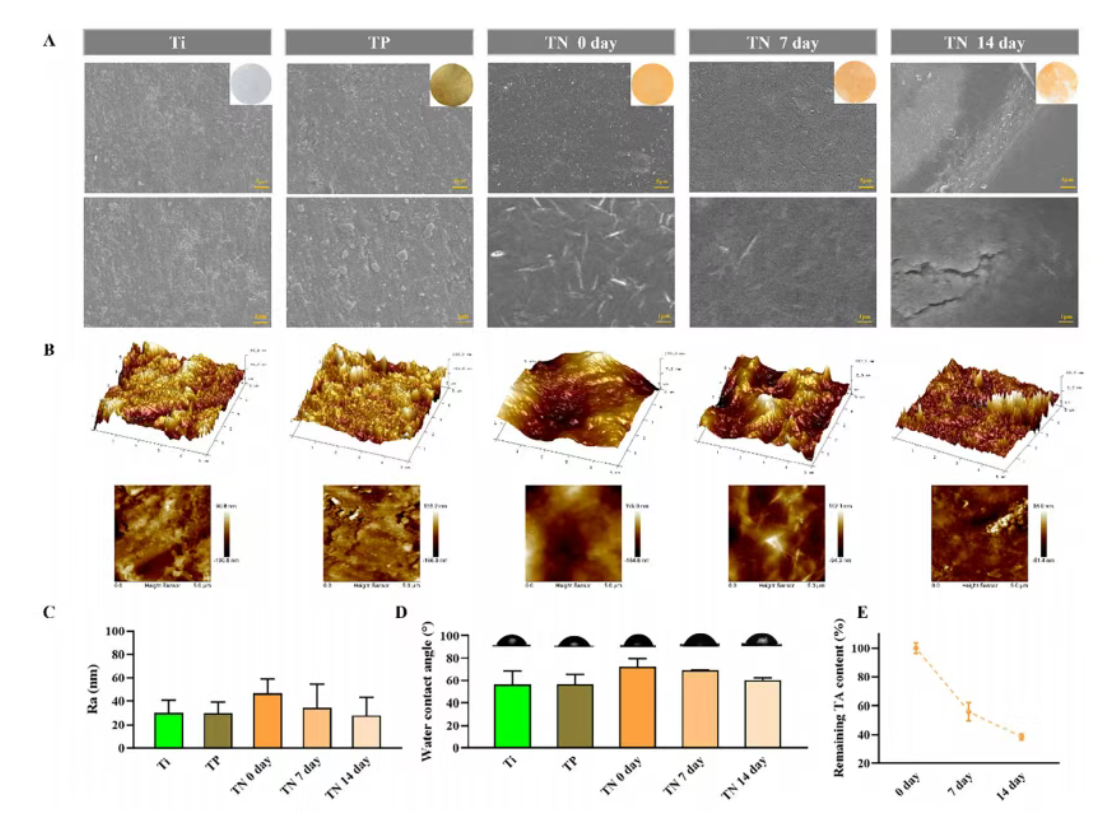
Figure 3. (A) Scanning electron microscopy images and corresponding digital photographs of Ti, Ti&PDA (TP), and Ti&PDA@NRs (TN) surfaces, including TN after immersion in artificial saliva for 0, 7, and 14 days. (B) Representative atomic force microscopy images and (C) corresponding surface roughness (Ra) values. (D) Water contact angle measurements of Ti, TP, and TN surfaces after immersion in artificial saliva for 0, 7, and 14 days. (E) Remaining tanshinone IIA content on TN surfaces determined by HPLC after immersion in artificial saliva for 0, 7, and 14 days.
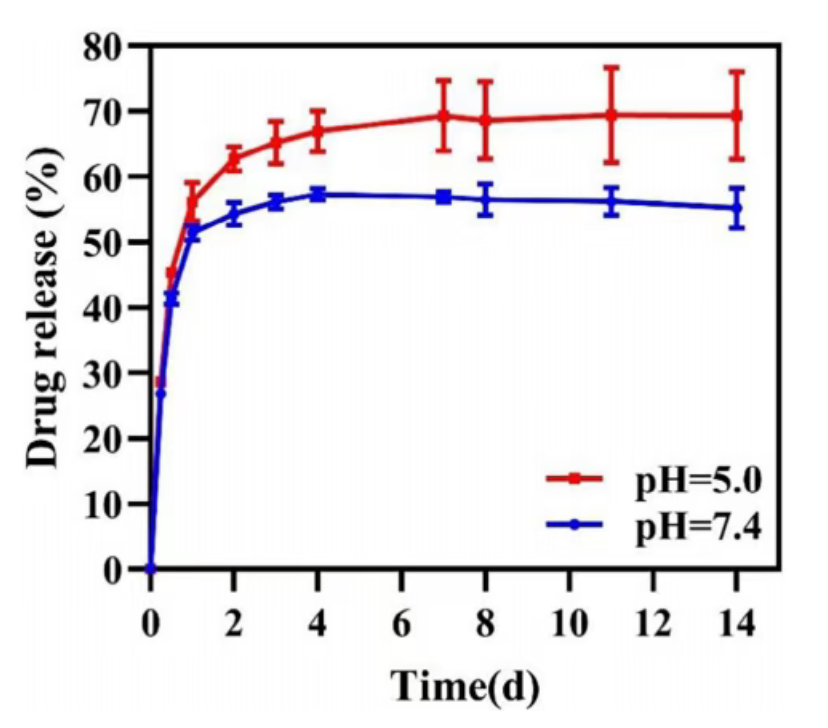
Figure 4. Release profiles of TA from the Ti&PDA@NRs coating under pH 5.0 and pH 7.4 conditions.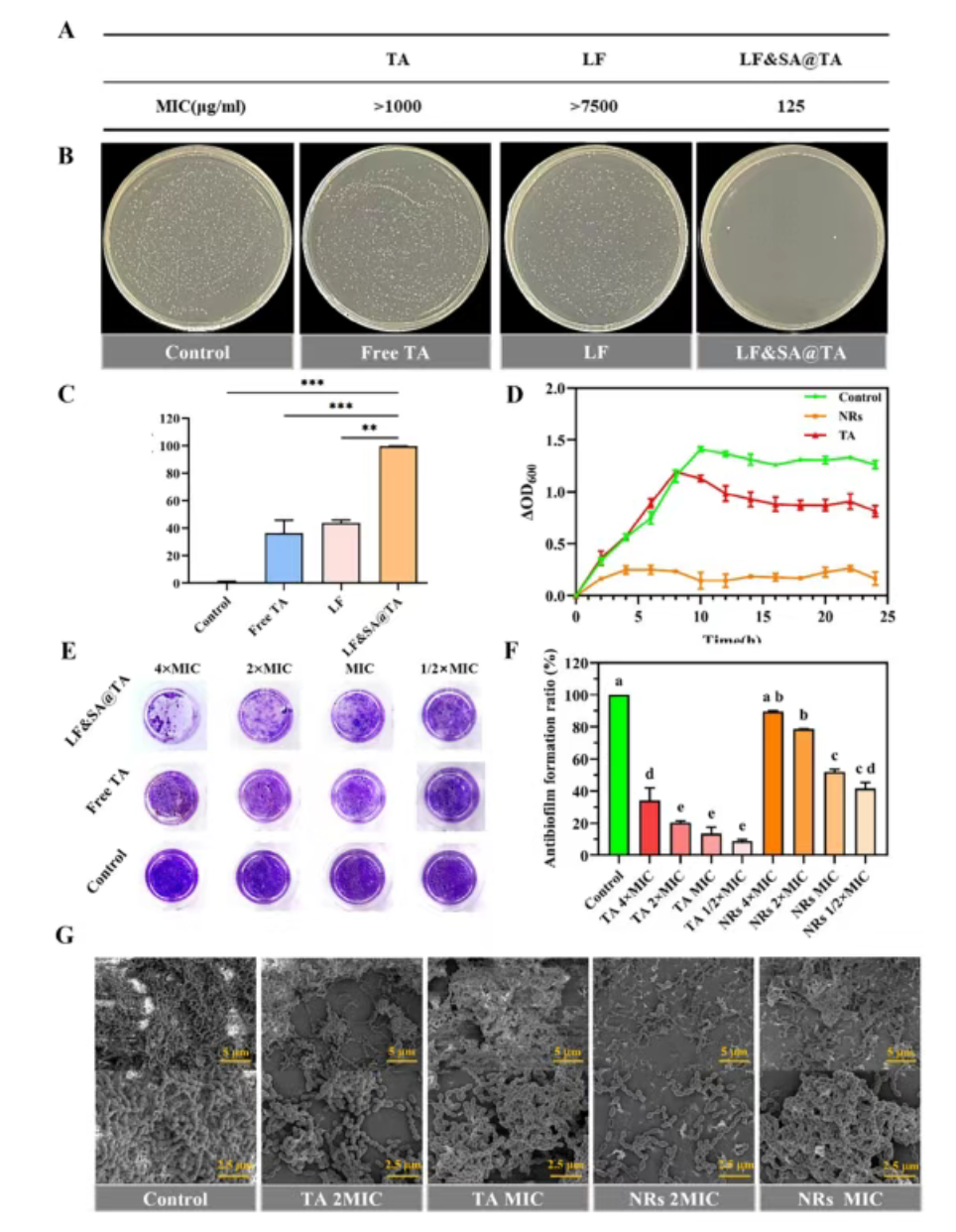
Figure 5. (A) Minimum inhibitory concentration values of different antibacterial agents against Streptococcus mutans. (B) Representative images of S. mutans colonies on BHI agar plates for each treatment condition and corresponding inhibition rates. (C) Statistical analysis of S. mutans colony counts in different treatment groups. (D) Growth curves of S. mutans over time in culture media containing antibacterial agents. Control group used PBS; treatment groups used free tanshinone IIA (125 µg/mL) or LF&SA@TA nanorods (containing an equivalent tanshinone IIA concentration of 125 µg/mL). (E) Crystal violet staining images of biofilms after treatment with PBS, free tanshinone IIA, or LF&SA@TA nanorods. (F) Quantitative assessment of biofilm biomass based on crystal violet staining, expressed as anti-biofilm formation rate relative to the control group. (G) Scanning electron microscopy images of biofilm structure morphology after 24 hours of treatment. Abbreviation “MIC” indicates the concentration of the test sample; both free tanshinone IIA and LF&SA@TA nanorods were applied at a concentration of 125 µg/mL.
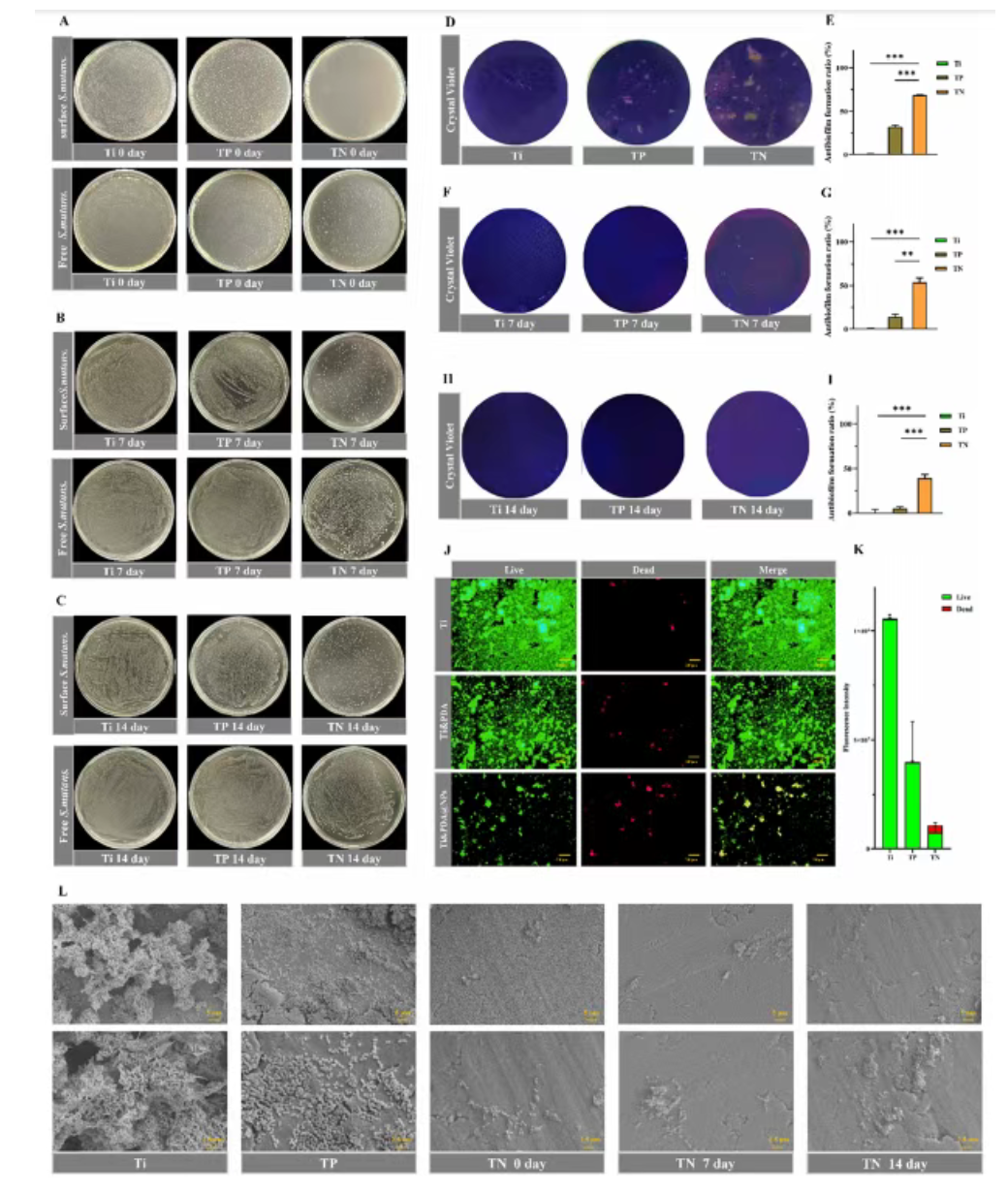
Figure 6. Antibacterial and anti-biofilm activity of Ti&PDA@NRs coating against Streptococcus mutans. (A-C) Representative photographs of colony-forming unit assays for surface adherent and planktonic S. mutans on Ti, Ti&PDA, and Ti&PDA@NRs surfaces after immersion in artificial saliva for 0, 7, and 14 days. (D-I) Crystal violet staining images of S. mutans biofilms formed on Ti, Ti&PDA, and Ti&PDA@NRs surfaces after immersion in artificial saliva for 0, 7, and 14 days, along with corresponding quantitative analysis results. (J) Live/dead fluorescence staining images of S. mutans on Ti, Ti&PDA, and Ti&PDA@NRs surfaces (green fluorescence indicates live bacteria, red fluorescence indicates dead bacteria). (K) Quantitative analysis of live/dead fluorescence intensity corresponding to (J). (L) Scanning electron microscopy images of S. mutans adhesion on Ti, Ti&PDA, and Ti&PDA@NRs surfaces after immersion in artificial saliva for 0, 7, and 14 days.
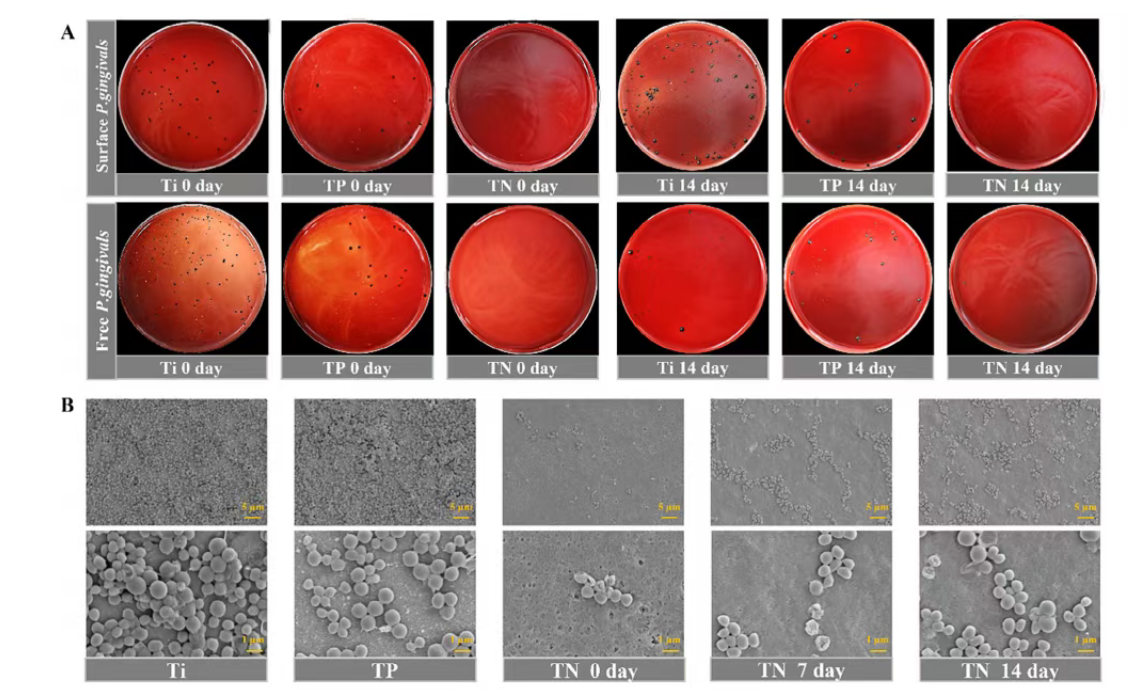
Figure 7. Antibacterial and anti-biofilm activity of Ti&PDA@NRs coating against Porphyromonas gingivalis. (A) Representative CFU culture dish images showing the growth of P. gingivalis on Ti, Ti&PDA, and Ti&PDA@NRs surfaces after immersion in artificial saliva for 0 and 14 days. These CFU culture dish images were used to qualitatively evaluate the contact killing effect on surface adherent bacteria and the release killing effect on planktonic bacteria. (B) Representative scanning electron microscopy images showing the adhesion of P. gingivalis on Ti, Ti&PDA, and Ti&PDA@NRs surfaces after immersion in artificial saliva for 0, 7, and 14 days. Dense bacterial aggregates were observed on Ti and Ti&PDA surfaces, while the Ti&PDA@NRs coating showed reduced bacterial attachment at all time points, with only a slight increase in adhesion after prolonged immersion.
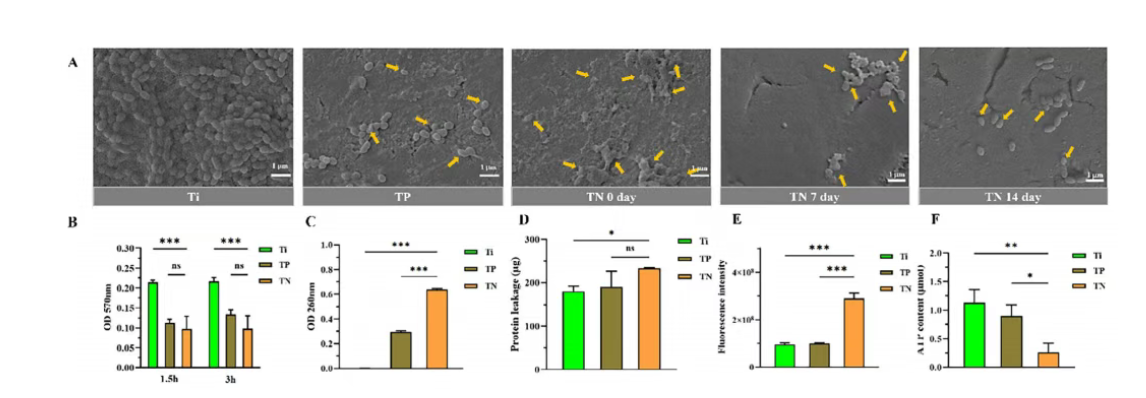
Figure 8. Mechanistic study data on the antibacterial effect of Ti&PDA@NRs coating against Streptococcus mutans. (A) Scanning electron microscopy images of S. mutans on Ti, polydopamine modified Ti, and Ti&PDA@NRs coating surfaces after immersion in artificial saliva for 0, 7, and 14 days, visually reflecting the state of bacterial adhesion on different material surfaces over time. (B) Sodium dodecyl sulfate sensitivity assay evaluating bacterial cell membrane integrity; results showed that bacteria treated with the coating exhibited significantly enhanced sensitivity to the detergent, indicating compromised cell membrane barrier function. (C) and (D) Quantitative measurements of DNA and protein leakage from S. mutans after culture on different material surfaces; the experimental data directly confirm that coating treatment leads to increased bacterial cell membrane permeability and leakage of intracellular contents. (E) Quantitative analysis of intracellular reactive oxygen species levels in S. mutans induced by coating treatment, measured using a fluorescent probe; data showed significantly higher fluorescence intensity in the treatment group compared to the control group, indicating that the coating triggers a strong oxidative stress response. (F) Measurement of intracellular ATP content in S. mutans after 24 hours of incubation on different material surfaces; results showed a sharp decrease in ATP content in bacteria on the Ti&PDA@NRs coating surface, clearly indicating that the antibacterial action of the coating also involves severe interference with bacterial energy metabolism. These experiments systematically reveal the complete pathway by which the coating achieves efficient antibacterial effects through multiple synergistic mechanisms, including cell structure disruption, content leakage, oxidative stress, and energy metabolism inhibition.
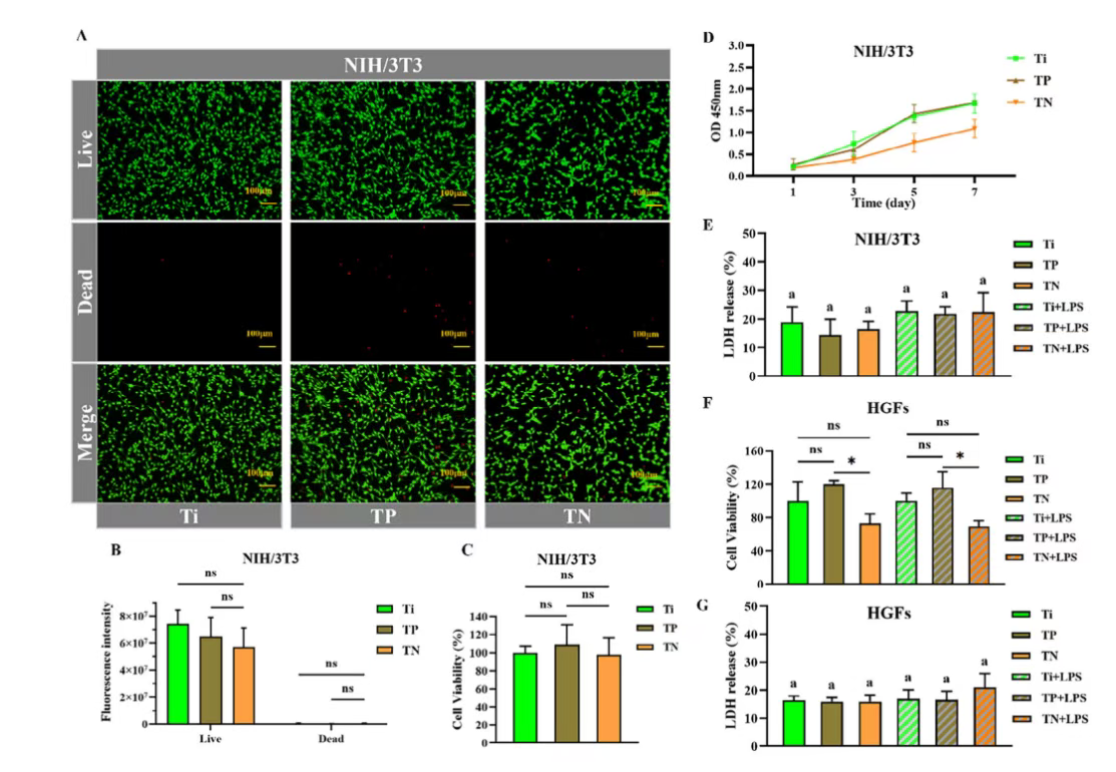
Figure 9. In vitro cytocompatibility evaluation of Ti, Ti&PDA (TP), and Ti&PDA@NRs (TN). (A) Live/dead fluorescence staining of NIH/3T3 cells after 24 hours of culture on different surfaces (live cells: green, dead cells: red). (B) Quantitative analysis of live/dead fluorescence intensity. (C) Relative cell viability of NIH/3T3 cells after 24 hours of culture, determined by CCK 8 assay and normalized to the Ti control group. (D) Metabolic activity of NIH/3T3 cells cultured on different surfaces for 1, 3, 5, and 7 days, assessed by CCK 8 assay. (E) Lactate dehydrogenase (LDH) release from NIH/3T3 cells under normal and inflammatory conditions induced by lipopolysaccharide from Porphyromonas gingivalis (LPS-Pg). (F) Relative viability of HGFs cultured on different surfaces under normal and inflammatory conditions, determined by CCK 8 assay. (G) LDH release from HGFs under normal and inflammatory conditions.
Conclusion
In this study, a pH modulated coating based on natural antibacterial compounds was successfully developed for titanium abutment surfaces. Through synergistic electrostatic adsorption and EDC/NHS promoted covalent amide bonding, natural compound nanoparticles with pH dependent charge reversal properties were immobilized onto the PDA modified surface. The coating exhibited acid accelerated yet sustained TA release characteristics, with a considerable proportion of TA remaining stably on the coating surface over time. Consequently, the coating demonstrated sustained antibacterial and anti biofilm activity against Streptococcus mutans and Porphyromonas gingivalis over 0, 7, and 14 days. Mechanistic studies indicated that the antibacterial effect involves multiple complementary pathways, including membrane disruption, leakage of cellular contents, induction of oxidative stress, and metabolic dysfunction, which collectively mediate contact mediated and release mediated antibacterial effects, particularly notable in the early stages. In vitro evaluations further confirmed that the coating exhibited good cytocompatibility with fibroblasts under both normal and inflammatory conditions. Overall, this study presents a practical and bio inspired strategy for constructing antibacterial coatings on titanium abutments, highlighting its potential for preventing infections in the peri implant mucosal region and providing a translational framework for abutment surface functionalization.
Full Text Link: https://doi.org/10.1002/adhm.202504686
